
1,4 - Dioxin
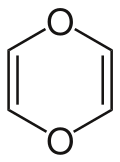
1,4-Dioxin is an organic compound with the chemical formula C4H4O2. It is a heterocyclic compound containing a six-membered ring with two oxygen atoms opposite each other. The compound is rarely encountered in its pure form but serves as a structural basis for a class of related compounds, including 1,4-dioxane, which is more commonly known and used. 1,4-Dioxin itself is unstable and prone to polymerization. It is of interest primarily in theoretical studies of aromaticity and in organic chemistry.
Definition and Structure
1,4-Dioxin is a heterocyclic organic compound with the chemical formula C₄H₄O₂. It consists of a six-membered ring containing four carbon atoms and two oxygen atoms, which are located at the 1 and 4 positions of the ring, hence the name 1,4-dioxin. The molecule is characterized by its oxygen atoms being opposite each other in the ring, creating a relatively planar structure. 1,4-Dioxin is the parent compound of a class of compounds known as dioxins, which include various chlorinated derivatives that are environmental pollutants.
Historical Background
The compound 1,4-dioxin itself is not as well-known or as widely discussed as its derivatives, particularly the chlorinated dioxins like 2,3,7,8-tetrachlorodibenzo-p-dioxin (TCDD). The recognition of dioxins as environmental contaminants emerged in the mid-20th century, particularly after the discovery of their presence in herbicides like Agent Orange used during the Vietnam War. While 1,4-dioxin is more of a theoretical structure in organic chemistry, its derivatives have been the focus of significant research due to their toxicological and environmental impacts.
Chemical Properties
1,4-Dioxin is a relatively stable organic compound, though it is more reactive than its isomer, 1,2-dioxin, due to the positions of the oxygen atoms in the ring. The compound is moderately soluble in water and highly soluble in organic solvents. It is prone to undergo electrophilic substitution reactions and can form various derivatives, particularly through halogenation. The planar structure of 1,4-dioxin allows it to engage in π-π interactions, which can influence its chemical behavior in complex environments. However, pure 1,4-dioxin is less commonly encountered compared to its more toxic chlorinated analogs.
Synthesis and Production
The synthesis of 1,4-dioxin typically involves the oxidation of suitable precursors, such as 1,4-butanediol, under controlled conditions. However, the synthesis and study of 1,4-dioxin are mainly of academic interest, as the compound itself is not commonly used in industry. The focus of industrial and environmental concerns is more on the chlorinated dioxins, which are by-products of various chemical processes, including the manufacture of herbicides, paper bleaching, and waste incineration. These processes can inadvertently produce dioxins, leading to environmental contamination.
Applications
1,4-Dioxin, in its pure form, has limited direct applications. It is primarily studied as a chemical intermediate or as a structure of interest in organic chemistry. The real concern with dioxins lies in their chlorinated derivatives, which have no intentional industrial uses but are produced as by-products in various processes. These chlorinated dioxins are highly toxic and persistent environmental pollutants, known for their potential to cause cancer, reproductive and developmental problems, and other serious health issues.
Agricultural Uses
1,4-Dioxin itself is not used in agriculture, but its derivatives, particularly chlorinated dioxins, have been historically linked to agricultural chemicals. The most infamous example is the contamination of herbicides, such as Agent Orange, with TCDD, a highly toxic dioxin. This contamination led to severe environmental and health consequences in areas where these herbicides were used. The use of chlorinated dioxins in agriculture is now heavily regulated or banned due to their persistence in the environment and their ability to bioaccumulate in the food chain.
Non-Agricultural Uses
Non-agricultural uses of 1,4-dioxin are minimal due to its limited practical applications. However, its chlorinated derivatives have been studied extensively due to their presence as contaminants in various industrial processes. These include the production of certain pesticides, the bleaching of paper pulp using chlorine, and the incineration of waste materials. The unintentional formation of dioxins during these processes has led to significant environmental and health concerns, prompting efforts to minimize dioxin emissions and contamination.
Health Effects
1,4-Dioxin itself is not widely recognized as a significant health threat, but its chlorinated derivatives are among the most toxic environmental pollutants known. Chlorinated dioxins, such as TCDD, are classified as human carcinogens and have been linked to a variety of health issues, including immune system suppression, endocrine disruption, and reproductive and developmental problems. These compounds can persist in the environment for decades, leading to long-term exposure risks for humans and wildlife. The toxicity of these compounds is due to their ability to bind to the aryl hydrocarbon receptor (AhR) in cells, leading to a cascade of harmful biological effects.
Human Health Effects
Human exposure to chlorinated dioxins, such as those related to 1,4-dioxin, can occur through contaminated food, air, water, and soil. These compounds are lipophilic, meaning they accumulate in fatty tissues and can persist in the human body for extended periods. Long-term exposure has been associated with an increased risk of various cancers, particularly in the liver, lungs, and skin. Other health effects include developmental and reproductive issues, such as birth defects and reduced fertility, as well as immune system dysfunction. The World Health Organization (WHO) and other regulatory bodies have set strict limits on dioxin exposure to minimize health risks.
Environmental Impact
The environmental impact of 1,4-dioxin is primarily of concern when considering its chlorinated derivatives. These dioxins are highly persistent in the environment, resisting degradation and accumulating in the food chain. They can be transported over long distances in the atmosphere, leading to widespread contamination. Dioxins are particularly harmful to aquatic ecosystems, where they can accumulate in sediments and enter the food web, affecting fish, birds, and mammals. The long-term persistence and bioaccumulation of dioxins make them a significant environmental hazard, requiring careful monitoring and management to prevent contamination.
Regulation and Guidelines
Due to the severe health and environmental risks associated with chlorinated dioxins, including those related to 1,4-dioxin, strict regulations have been established globally. The Stockholm Convention on Persistent Organic Pollutants (POPs) is an international treaty aimed at reducing and eliminating the production and release of dioxins. In the United States, the Environmental Protection Agency (EPA) regulates dioxin emissions and sets maximum allowable concentrations in the environment. Similar regulations exist in the European Union and other regions, focusing on reducing dioxin formation during industrial processes and monitoring dioxin levels in food, water, and air.
Controversies and Issues
The controversies surrounding 1,4-dioxin and its derivatives primarily stem from their environmental persistence and toxicological effects. The use of dioxin-contaminated herbicides during the Vietnam War led to widespread health issues, sparking debates over the responsibilities of governments and corporations in addressing the fallout. Additionally, the continued presence of dioxins in the environment, despite regulatory efforts, raises concerns about the effectiveness of current pollution control measures. The disposal of dioxin-contaminated waste and the remediation of contaminated sites remain contentious issues, with ongoing discussions about the best approaches to mitigate the impact of these persistent pollutants.
Treatment Methods
Treating contamination from dioxins, including those related to 1,4-dioxin, involves several approaches to remove or neutralize these compounds in the environment. One method is thermal treatment, such as incineration at high temperatures, which can break down dioxins into less harmful substances. However, this process must be carefully controlled to prevent the formation of new dioxins. Chemical methods, such as using reagents to degrade dioxins, are also employed, though they can be expensive and complex. Bioremediation, which uses microorganisms to degrade dioxins, is an emerging method showing promise for treating contaminated soils and sediments. Phytoremediation, using plants to absorb and break down dioxins, offers a more sustainable and less invasive approach.
Monitoring and Testing
Monitoring and testing for 1,4-dioxin and its derivatives are critical for assessing environmental contamination and exposure risks. Analytical methods such as gas chromatography coupled with mass spectrometry (GC-MS) are the standard techniques for detecting and quantifying dioxins in environmental samples, including air, water, soil, and biological tissues. Regular monitoring of dioxin levels is essential for ensuring compliance with regulatory standards and for tracking the effectiveness of pollution control measures. Advances in analytical technologies continue to improve the sensitivity and accuracy of dioxin detection, supporting better management and mitigation of dioxin-related risks.
References
- ATSDR (Agency for Toxic Substances and Disease Registry). (2020). Beryllium Toxicity. Retrieved from https://www.atsdr.cdc.gov/
- EPA (Environmental Protection Agency). (2020). 2,4-Dichlorophenoxyacetic Acid (2,4-D). Retrieved from https://www.epa.gov/
- EPA (Environmental Protection Agency). (2021). Home Water Treatment Units. Retrieved from https://www.epa.gov/
- IARC (International Agency for Research on Cancer). (2015). 2,4-Dichlorophenoxyacetic Acid (2,4-D). Retrieved from https://www.iarc.fr/
- WHO (World Health Organization). (2011). Guidelines for Drinking-Water Quality. Retrieved from https://www.who.int/
p-dioxin
( C4H4O2 )
| Parameter | Details |
|---|---|
| Source | Industrial processes, solvent use, byproduct of chemical manufacturing |
| MCL | No specific MCL; EPA has a health advisory level at 0.35 µg/L |
| Health Effects | Potential carcinogen, liver and kidney damage, respiratory issues |
| Detection | GC-MS, liquid-liquid extraction |
| Treatment | Advanced oxidation processes, activated carbon |
| Regulations | US EPA advisory, state regulations |
| Monitoring | Regular testing near industrial sites and areas of concern |
| Environmental Impact | Soil and water contamination, persistent in the environment |
| Prevention | Proper disposal, use of safer alternatives |
| Case Studies | Contamination incidents in industrial areas |
| Research | Health impacts, improved detection and remediation methods |
Other Chemicals in Water
1,4-Dioxin in drinking water
| Property | Value |
|---|---|
| Preferred IUPAC Name | 1,4-Dioxin |
| Other Names | p-Dioxin, Dioxane |
| CAS Number | 123-91-1 |
| Chemical Formula | C4H8O2 |
| Molar Mass | 88.11 g/mol |
| Appearance | Colorless liquid |
| Melting Point | 11.8 °C (53.2 °F) |
| Boiling Point | 101 °C (214 °F) |
| Solubility in Water | Fully miscible |
Videos

